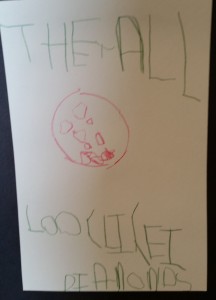Incorporating science vocabulary, sketching, and writing with science experiences is a natural progression in our classroom. While wishing and waiting for winter to truly arrive, we decided to introduce the idea of crystals to the children. We started by exploring crystals such as polyacrylamide, a super absorbent type that can be found in disposable diapers. The children each received a petri dish containing a small amount of the substance, as well as a cup of water and a pipette.
As they dripped water onto the tiny crystals, they noticed that the crystals absorbed the water. We also discussed that, even though the crystals grew and resembled ice, the crystals were inedible. The children became very excited as the crystals grew so large, they overflowed from the petri dishes and filled trays.
The children noted that the crystals were no longer tiny, hard, and dry (think table salt), but were now large, squishy, and wet.
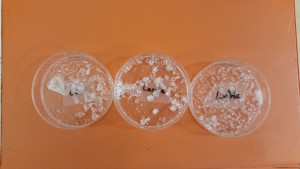
On another day, we gave the children another type of small, dry crystal…Alum. The children were again given a tiny amount in a petri dish. This time they added hot tap water and stirred. The crystals dissolved. Most thought that this was pretty boring, so we set them aside. However, on the following day, we checked the petri dishes. Wow:
The crystals had grown a little bigger, the water had evaporated, and each resulting group of crystals appeared a bit different. This presented a perfect opportunity to sketch and write about our findings:
We also incorporated the math concepts of sorting, classifying, and patterning with artificial crystals. The mirrored work surfaces highlighted their explorations.