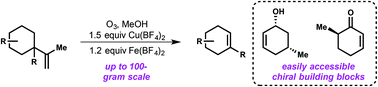Newhouse Group at Yale:
(56) Pengpeng Zhang, Timothy R. Newhouse*. “Palladium-Catalyzed Carbonylative Difunctionalization of Unactivated Alkenes Initiated by Unstabilized Enolates” Angew. Chem. Int. Ed. 2023, DOI: 10.1002/anie.202307455
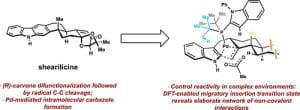
(55) Daria E. Kim, Yingchuan Zhu, Shingo Harada, Isaiah Aguilar, Abbigayle E. Cuomo, Minghao Wang, Timothy R. Newhouse*. “Total Synthesis of (+)-Shearilicine” J. Am. Chem. Soc. 2023, 8, 4394-4399. DOI: 10.1021/jacs.2c13584
(54) Abbigayle E. Cuomo, Sebastian Ibarran, Sanil Sreekumar, Haote Li, Jungmin Eun, Jan Paul Menzel, Pengpeng Zhang, Frederic Buono, Jinhua J. Song, Robert H. Crabtree, Victor S. Batista*, Timothy R. Newhouse*. “Feed-Forward Neural Network for Predicting Enantioselectivity of the Asymmetric Negishi Reaction” ACS Cent. Sci. 2023, DOI: 10.1021/acscentsci.3c00512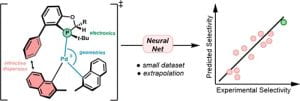
(53) Edward A. deRamon, Venkata R. Sabbasani, Matthew D. Streeter, Yannan Liu, Timothy R. Newhouse, David M. McDonald, David A. Spiegel*. “Pentosinane, a Post-Translational Modification of Human Proteins with Underappreciated Stability” J. Am. Chem. Soc. 2022, DOI: 10.1021/jacs.2c09626
(52) Alexandra K. Bodnar, Suzanne M. Szewczyk, Yang Sun, Yifeng Chen, Anson X. Huang, Timothy R. Newhouse. “A Comparative Mechanistic Analysis of Palladium- and Nickel-Catalyzed α,β-Dehydrogenation of Carbonyls via Organozinc Intermediates” ChemRxiv. Cambridge: Cambridge Open Engage; 2022, DOI: 10.26434/chemrxiv-2022-0t55g
(51) Jungmin Eun, Timothy R. Newhouse. “Computational decoding” Nature Reviews Chemistry, 2022, 6, 168-169.
(50) Yannan Liu, Alexander W. Schuppe, Yizhou Zhao, Jaehoo Lee, Timothy R. Newhouse. “Synthesis of (–)-melazolide B, a degraded limonoid, from a natural terpene precursor” Tetrahedron Chem. 1, 2022, DOI: 10.1016/j.tchem.2022.100011

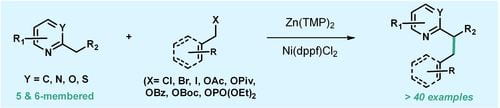
(49) Pengpeng Zhang, Jin Wang, Zoe R. Robertson, Timothy R. Newhouse. “Coordination-Controlled Nickel-Catalyzed Benzylic Allylation of Unactivated Electron-Deficient Heterocycles” Angew. Chem. Int. Ed. 2022, DOI: 10.1002/anie.202200602
(48) Pengpeng Zhang, Timothy R. Newhouse*. “Thiophene, 2-bromo-5-methyl-” Encyclopedia for Reagents in Organic Synthesis 2021, DOI: 10.1002/047084289X.rn02366
(47) Alexander W. Schuppe, Yannan Liu, Yizhou Zhao, Sebastian Ibarraran, David Huang, Emma Wang, Jaehoo Lee, Patrick Loria, Timothy R. Newhouse. “Strategies for the De Novo Synthesis of Highly Substituted Pyridine Scaffolds: Unified Total Synthesis of the Limonoid Alkaloids” ChemRxiv. Cambridge: Cambridge Open Engage; 2021.
(46) Pengpeng Zhang†, Jungmin Eun†, Masha Elkin, Yizhou Zhao, Rachel L. Cantrell, and Timothy R. Newhouse. “A Neural Network Model Informs Total Synthesis of Clovane Sesquiterpenoids” ChemRxiv. Cambridge: Cambridge Open Engage; 2021.
(45) Alexandra K. Bodnar, Aneta Turlik, David Huang, Will Butcher, Joanna K. Lew, Timothy R. Newhouse. “Preparation of Hindered Aniline CyanH and Application in the Allyl-Ni-Catalyzed α,β-Dehydrogenation of Carbonyls” Org. Synth. 2021, 98, 263-288. DOI: 10.15227/orgsyn.098.0263
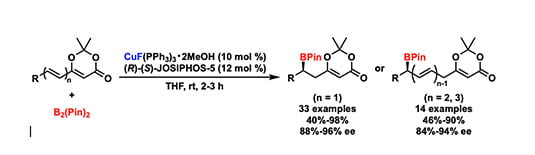
(44) Chang-Yun Shi†, Jungmin Eun†, Timothy R. Newhouse*, Liang Yin* “Copper(I)‐Catalyzed Asymmetric Conjugate 1,6‐, 1,8‐, and 1,10‐Borylation” Angew. Chem. Int. Ed. 2021, xx-xx. DOI: 10.1002/anie.202016081
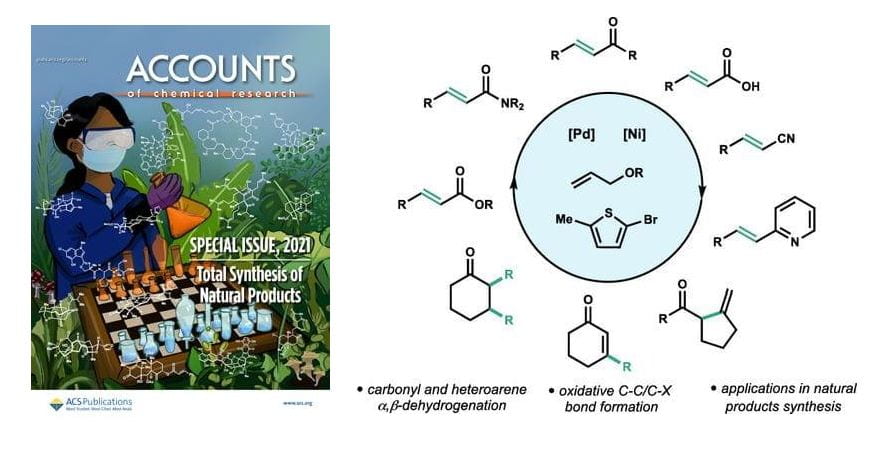
(43) David Huang†, Timothy R. Newhouse. “Dehydrogenative Pd and Ni Catalysis for Total Synthesis” Acc. Chem. Res. 2021, 54, 1118-1130. DOI: 10.1021/acs.accounts.0c00787
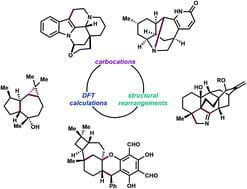
(42) Alexander W. Schuppe†, Yannan Liu†, Timothy R. Newhouse. “An Invocation for Computational Evaluation of Isomerization Transforms: Cationic Skeletal Reorganizations as a Case Study” Natural Product Reports, 2021, 38, 510-527. DOI: 10.1039/D0NP0005A
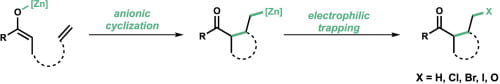
(41) Diego Olivieri†, David Huang†, Alexandra K. Bodnar, Shijin Yu, Timothy R. Newhouse. “Zinc-mediated anionic cyclization of unstabilized ketone enolates with unactivated alkenes” Tetrahedron 2020, 76. DOI: 10.1016/j.tet.2020.131417
- Featured in:
Org. Chem. Highlights: Metal-Mediated C-C Ring Construction
(40) Yannan Liu, Tina A. Holt, Andrei Kutateladze, Timothy R. Newhouse. “Stereochemical Revision of Xylogranatin F by GIAO and DU8+ NMR Calculations” Chirality, 2020, 32, 515-523. DOI: 10.1002/chir.23189 (39) Pengpeng Zhang, David Huang, Timothy R. Newhouse. “Aryl-Nickel-Catalyzed Benzylic Dehydrogenation of Electron-Deficient Heteroarenes” J. Am. Chem. Soc. 2020 142, 1757-1762. DOI:10.1021/jacs.9b12706
(39) Pengpeng Zhang, David Huang, Timothy R. Newhouse. “Aryl-Nickel-Catalyzed Benzylic Dehydrogenation of Electron-Deficient Heteroarenes” J. Am. Chem. Soc. 2020 142, 1757-1762. DOI:10.1021/jacs.9b12706
(38) Pengpeng Zhang, Timothy R. Newhouse. “Oxidation Stepping Stones: α-Oxytriflation Enables Asymmetric Arylation of Amides” Chem. 2019, 5(7), 1690-1692. DOI: https://doi.org/10.1016/j.chempr.2019.06.008
(37) David Huang, Diego Olivieri, Yang Sun, Pengpeng Zhang, Timothy R. Newhouse. “Nickel-Catalyzed Difunctionalization of Unactivated Alkenes Initiated by Unstabilized Enolates” J. Am. Chem. Soc. 2019, 141, 16249-16254. DOI:10.1021/jacs.9b09245
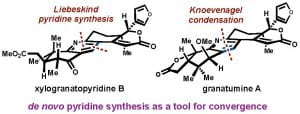
(36) Alexander W. Schuppe†, Yizhou Zhao†, Yannan Liu, Timothy R. Newhouse. “Total Synthesis of (+)-Granatumine A and Related Bislactone Limonoid Alkaloids via a Pyran to Pyridine Interconversion” J. Am. Chem. Soc. 2019, 141, 9191-9126. DOI:10.1021/jacs.9b04508

(35) Aneta Turlik, Yifeng Chen, Anthony C. Scruse, Timothy R. Newhouse. “Convergent Total Synthesis of Principinol D, a Rearranged Kaurane Diterpenoid” J. Am. Chem. Soc. 2019, 141, 8088-8092. DOI:10.1021/jacs.9b03751
(34) Joshua E. Zweig, Tongil A. Ko, Junrou Huang, Timothy R. Newhouse. “Effects of π-Extension on Pyrrole Hemithioindigo Photoswitches.” Tetrahedron. 2019, 75(34), 130466 DOI:10.1016/j.tet.2019.130466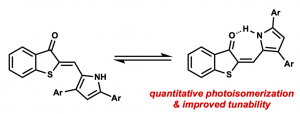
(33) David Huang, Suzanne M. Szewczyk, Pengpeng Zhang, Timothy R. Newhouse. “Allyl-Nickel Catalysis Enables Carbonyl Dehydrogenation and Oxidative Cycloalkenylation of Ketones” J. Am. Chem. Soc. 2019, 141, 5669–5674. DOI: 10.1021/jacs.9b02552.
-
- Featured in:

(32) Daria E. Kim, Joshua E. Zweig, Timothy R. Newhouse. “Total Synthesis of Paspaline A and Emindole PB Enabled by Computational Augmentation of a Transform-Guided Retrosynthetic Strategy” J. Am. Chem. Soc. 2019, 141, 1479–1483. DOI: 10.1021/jacs.8b13127.
-
- Featured in:
- C&EN News: Computation helps chemists shorten synthesis of natural product
- Chemistry World: Computation trick cuts natural product synthesis from 27 to nine steps
- YaleNews: Synthesizing natural products with a computational step saver
- Computational Chemistry Highlights: Computationally Augmented Retrosynthesis: Total Synthesis of Paspaline A and Emindole PB
- Synfacts: Synthesis of Paspaline A and Emindole PB
- Featured in:

(31) Masha Elkin†, Anthony C. Scruse†, Aneta Turlik, Timothy R. Newhouse. “Computational and Synthetic Investigation of Cationic Rearrangement in the Putative Biosynthesis of Justicane Triterpenoids” Angew. Chem. Int. Ed. 2019, 58, 1025–1029. DOI:10.1002/anie.201810566.
(30) Daria E. Kim, Yingchuan Zhu, Timothy R. Newhouse. “Vinylogous acyl triflates as an entry point to α,β-disubstituted cyclic enones via Suzuki–Miyaura cross-coupling” Org. Biomol. Chem. 2019, 17, 1796–1799. DOI: 10.1039/c8ob02573h.

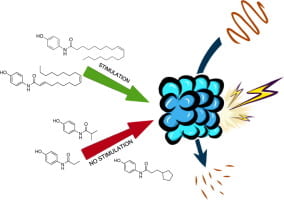
(29) Rachel A. Coleman, Christine S. Muli, Yizhou Zhao, Atul Bhardwaj, Timothy R. Newhouse, Darci J. Trader. “Analysis of chain length, substitution patterns, and unsaturation of AM-404 derivatives as 20S proteasome stimulators” Bioorganic Med. Chem. Lett. 2019, 29, 420–423. DOI:10.1016/j.bmcl.2018.12.030
(28) Masha Elkin, Timothy R. Newhouse. “Computational chemistry strategies in natural product synthesis” Chem. Soc. Rev. 2018, 47, 7830–7844. DOI: 10.1039/C8CS00351C
-
- Cover feature
 |
 |
(27) Hai-Jun Zhang, Alexander W. Schuppe, Shi-Tao Pan, Jin-Xiang Chen, Bo-Ran Wang, Timothy R. Newhouse,* and Liang Yin.* “Copper-Catalyzed Vinylogous Aerobic Oxidation of Unsaturated Compounds with Air” J. Am. Chem. Soc. 2018, 140, 5300-5310. DOI: 10.1021/jacs.8b01886.

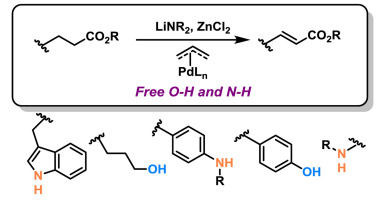
(26) Suzanne M. Szewczyk, Yizhou Zhao, Holt Sakai, Pascal Dube, Timothy R. Newhouse. “α,β-Dehydrogenation of esters with free O-H and N-H functionalities via allyl-palladium catalysis” Tetrahedron 2018, 74, 3293-3300. DOI: 10.1016/j.tet.2018.02.028.
(25) Alexander W. Schuppe, David Huang, Yifeng Chen, Timothy R. Newhouse. “Total Synthesis of (–)-Xylogranatopyridine B via a Palladium-Catalyzed Oxidative Stannylation of Enones” J. Am. Chem. Soc. 2018, 140, 2062-2066. DOI: 10.1021/jacs.7b13189.
-
- Featured in Synfacts

- Featured in Synfacts
(24) David Huang, Yizhou Zhao, Timothy R. Newhouse. Synthesis of Cyclic Enones by Allyl-Palladium-Catalyzed α,β-Dehydrogenation” Org. Lett. 2018, 20, 684–687. DOI: 10.1021/acs.orglett.7b03818.
-
- Featured inSynfacts

- Featured inSynfacts
(23) Yizhou Zhao, Yifeng Chen, Timothy R. Newhouse. “Allyl-Palladium Catalyzed α,β-Dehydrogenation of Carboxylic Acids via Enediolates” Angew. Chem. Int. Ed. 2017, 56, 13122-13125. DOI: 10.1002/anie.201706893.
-
- Featured in Synfacts

- Featured in Synfacts
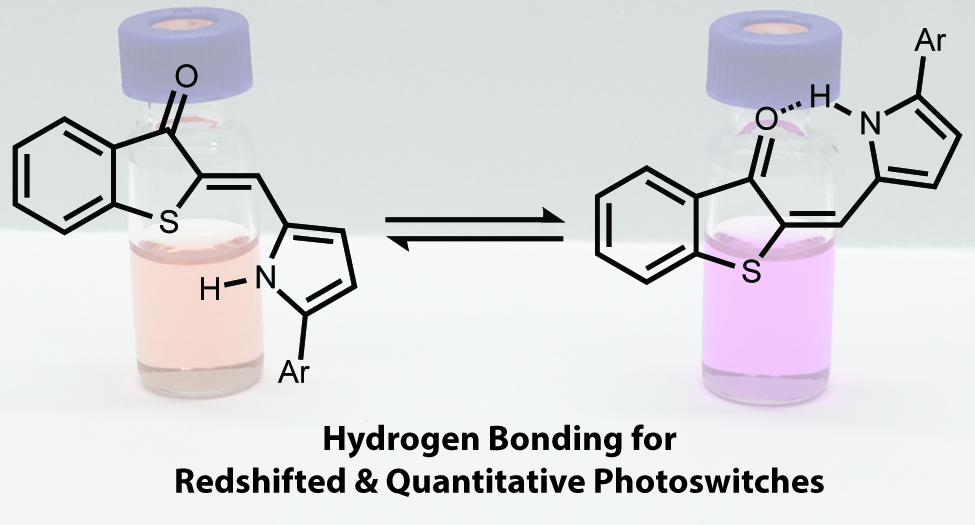
(22) Joshua E. Zweig, Timothy R. Newhouse. “Isomer-Specific Hydrogen Bonding as a Design Principle for Bidirectionally Quantitative and Redshifted Hemithioindigo Photoswitches” J. Am. Chem. Soc. 2017, 139, 10956-10959. DOI: 10.1021/jacs.7b04448.
(21) Gong Xu, Masha Elkin, Dean Tantillo, Timothy R. Newhouse,* Thomas Maimone.* “Traversing Biosynthetic Carbocation Landscapes in the Total Synthesis of Andrastin and Terretonin Meroterpenes” Angew. Chem. Int. Ed. 2017, 56, 12498-12502. DOI: 10.1002/anie.201705654.
-
- Featured in Organometallic Chemistry

- Featured in Organometallic Chemistry
(20) Yifeng Chen, David Huang†, Yizhou Zhao†, Timothy R. Newhouse. “Allyl-Palladium Catalyzed Ketone Dehydrogenation Enables Telescoping with Enone α,β-Vicinal Difunctionalization” Angew. Chem. Int. Ed. 2017, 56, 8258-8262. DOI: 10.1002/anie.201704874
(19) Joshua E. Zweig, Daria E. Kim, Timothy R. Newhouse. “Methods Utilizing First-Row Transition Metals in Natural Product Total Synthesis” Chem. Rev. 2017, 117, 11680-11752. DOI: 10.1021/acs.chemrev.6b00833.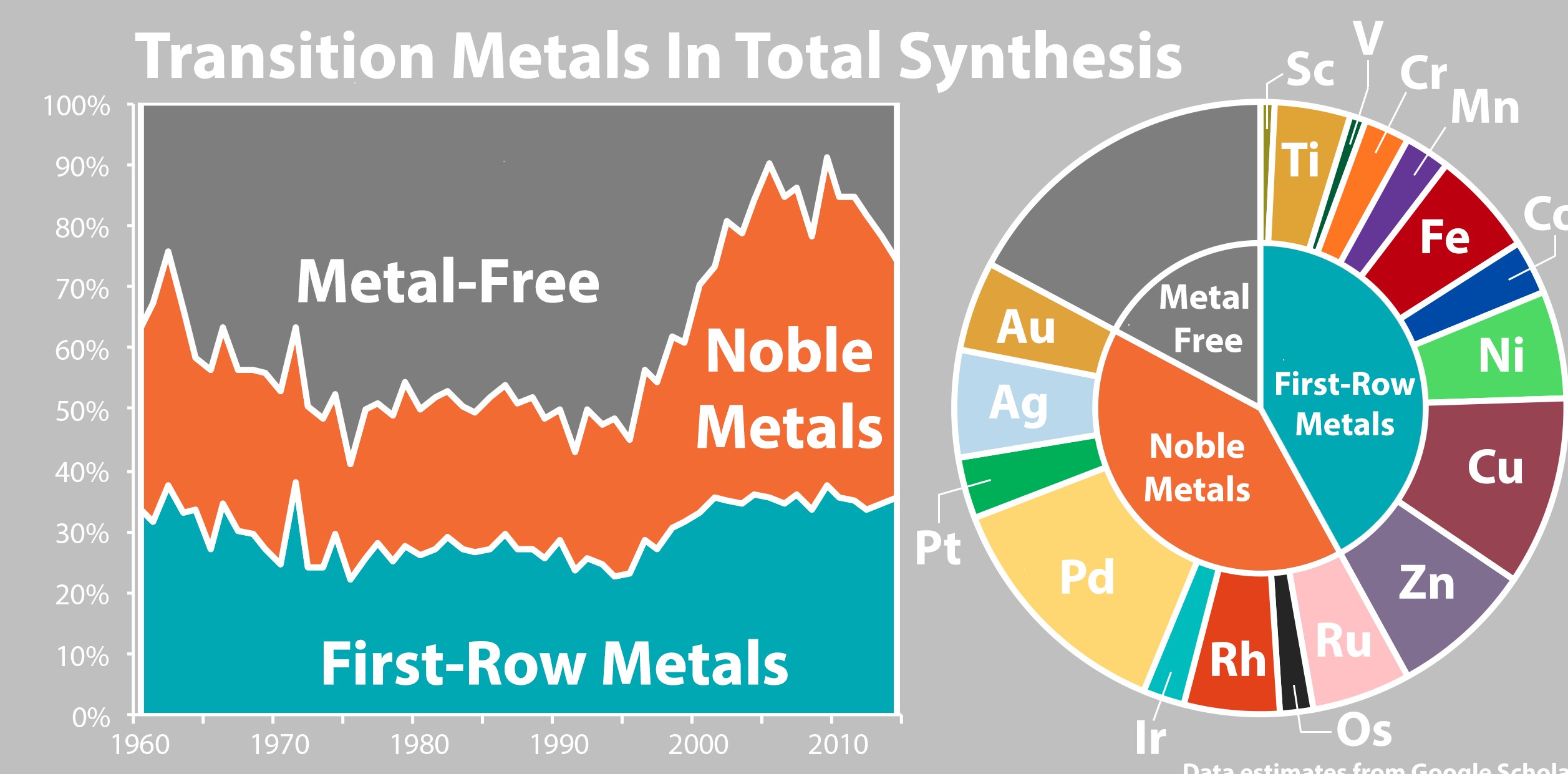
(18) Alexander W. Schuppe, James M. Cabrera, Catherine L. B. McGeoch, Timothy R. Newhouse.* “Scalable synthesis of enaminones utilizing Gold’s reagents” Tetrahedron 2017, 73, 3643–3651. DOI: 10.1016/j.tet.2017.03.092.
(17) Masha Elkin, Suzanne M. Szewczyk, Anthony C.Scruse, Timothy R. Newhouse. “Total Synthesis of (±)-Berkeleyone A” J. Am. Chem. Soc. 2017, 139, 1790-1793. DOI:10.1021/jacs.6b12914.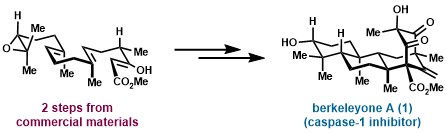
(16) Alexander W. Schuppe, Timothy R. Newhouse. “Assembly of the Limonoid Architecture by a Divergent Approach: Total Synthesis of (±)-Andirolide N via (±)-8α-Hydroxycarapin” J. Am. Chem. Soc. 2017, 139, 631-634. DOI: 10.1021/jacs.6b12268.
-
- Featured in Synfacts
(15) David Huang, Alexander W. Schuppe, Michael Z. Liang and Timothy R. Newhouse. “Scalable procedure for the fragmentation of hydroperoxides mediated by copper and iron tetrafluoroborate salts.” Org. Biomol. Chem. 2016, 14, 6197. DOI: 10.1039/c6ob00941g.
-
- This article is part of themed collection: Contemporary Synthetic Chemistry in Drug Discovery
(14) Yifeng Chen, Aneta Turlik, Timothy R. Newhouse. “Amide α,β-Dehydrogenation Using Allyl-Palladium Catalysis and a Hindered Monodentate Anilide.” J. Am. Chem. Soc. 2016, 138,1166. DOI: 10.1021/jacs.5b12924.
-
- Featured in Synfacts
(13) Aneta Turlik, Yifeng Chen, Timothy R. Newhouse. “Dehydrogenation Adjacent to Carbonyls Using Pd-Allyl Intermediates.” Synlett 2016, 27, 331. DOI: 10.1055/s-0035-1561282.

(12) Yifeng Chen, Justin P. Romaire, Timothy R. Newhouse. “Palladium-Catalyzed α,β-Dehydrogenation of Esters and Nitriles.” J. Am. Chem. Soc. 2015, 137, 5875. DOI:10.1021/jacs.5b02243.
-
- Featured in OPRD’s “Some Items of Interest to Process R&D Chemists and Engineers
TRN before Yale:
(11) T. R. Newhouse, P. Kaib, A. Gross, E. J. Corey. “Versatile Approaches for the Synthesis of Chiral Fused-Ring g-Lactones Utilizing Cyclopropane Intermediates.” Org. Lett. 2013, 15, 1591–1593.
(10) T. R. Newhouse, X. Li, M. M. Blewett, C. M. C. Whitehead, E. J. Corey. “A Tetradentate Ligand for the Enantioselective Ti(IV)-Promoted Oxidation of Sulfides to Sulfoxides: Origin of Enantioselectivity.” J. Am. Chem. Soc. 2012, 134, 17354 – 17357.
(9) L. Zou, R. S. Paton, A. Eschenmoser, T. R. Newhouse, P. S. Baran, K. N. Houk. “Enhanced Reactivity in Dioxirane C-H Oxidations via Strain Release.” J. Org. Chem. 2013, 78, DOI: 10.1021/jo400350v
(8) T. Newhouse, P. S. Baran. “If C-H Bonds Could Talk: Selective C-H Bond Oxidation.” Angew. Chem. Int. Ed. 2011, 50, 3362 – 3374.
(7) K. Foo, T. Newhouse, I. Mori, H. Takayama, P. S. Baran. “Total Synthesis Guided Structure Elucidation of (+)-Psychotetramine.” Angew. Chem. Int. Ed. 2011, 50, 2716 – 2719.
(6) T. Newhouse, C. A. Lewis, K. J. Eastman, P. S. Baran. “Scalable Total Syntheses of N-Linked Tryptamine Dimers by Direct Indole-Aniline Coupling: Psychotrimine and Kapakahines B and F.” J. Am.Chem. Soc. 2010, 132, 7119 – 7137.
(5) M. A. Schallenberger, T. Newhouse, P. S. Baran, F. E. Romesberg, “The Psychotrimine Natural Products Have Antibacterial Activity Against Gram-Positive Bacteria and Act Via Membrane Disruption.” J. Antibiot. 2010, 63, 685 – 687.
(4) T. Newhouse, P. S. Baran, R. W. Hoffmann. “The Economies of Synthesis.” Chem. Soc. Rev. 2009, 38, 3010 – 3021.
(3) T. Newhouse, C. A. Lewis, P. S. Baran. “Enantiospecific Total Syntheses of Kapakahines B and F.” J. Am. Chem. Soc. 2009, 131, 6360 – 6361.
(2) T. Newhouse, P. S. Baran. “Total Synthesis of (±)-Psychotrimine.” J. Am. Chem. Soc. 2008, 130, 10886 – 10887.
(1) R. I. Goldstein, R. Guo, C. Hughes, D. P. Maurer, T. Newhouse, T. J. Sisto, R. R. Conry, S. L. Price, D. M. Thamattoor. “Concomitant conformational dimorphism in 1,2-bis(9-anthryl)acetylene” CrystEngComm 2015, 17, 4877 – 4882.